Integrated Analytical and Stability Solutions for Product Development
The journey of transforming a pharmaceutical compound from discovery to commercial production is one of the most difficult processes in the life sciences sector. Although early-stage discoveries frequently show encouraging biological activity, converting these compounds into stable, manufacturable and legally compliant products necessitates a strong analytical and stability approach.
From preclinical studies to clinical trials, as drug candidates progress through development phases, pharmaceutical companies have to deliver dependable, repeatable and regulated analytical data that shows product quality, stability and safety and then ultimately suitable for commercial scale production. Strict quality criteria are met by active pharmaceutical ingredients (APIs), intermediate and final medicinal products through verification by analytical testing. At the same time, stability studies confirm that these final products remain secure and efficient throughout their specified self-life.
Before authorizing drugs, regulatory firms demand pharmaceutical organizations to provide reliable, validated analytical testing protocols, techniques and effective stability strategies. Drug manufacturers encounter holdups in authorization from regulatory bodies, more costly productions, data discrepancies in the absence of well-structured analytical techniques and stability initiatives and possible threats to product quality.
HPC Services for Analytical and Stability Studies
At HPC, we assist CDMOs, pharmaceutical firms and biotech pioneers in navigating this challenging path. Throughout the development and marketing of pharmaceuticals, our Analytical and Stability Services aim to produce trustworthy data. Our goal is simple to guide each molecule confidently through the development process with confidence, reliability and readiness for global regulations.
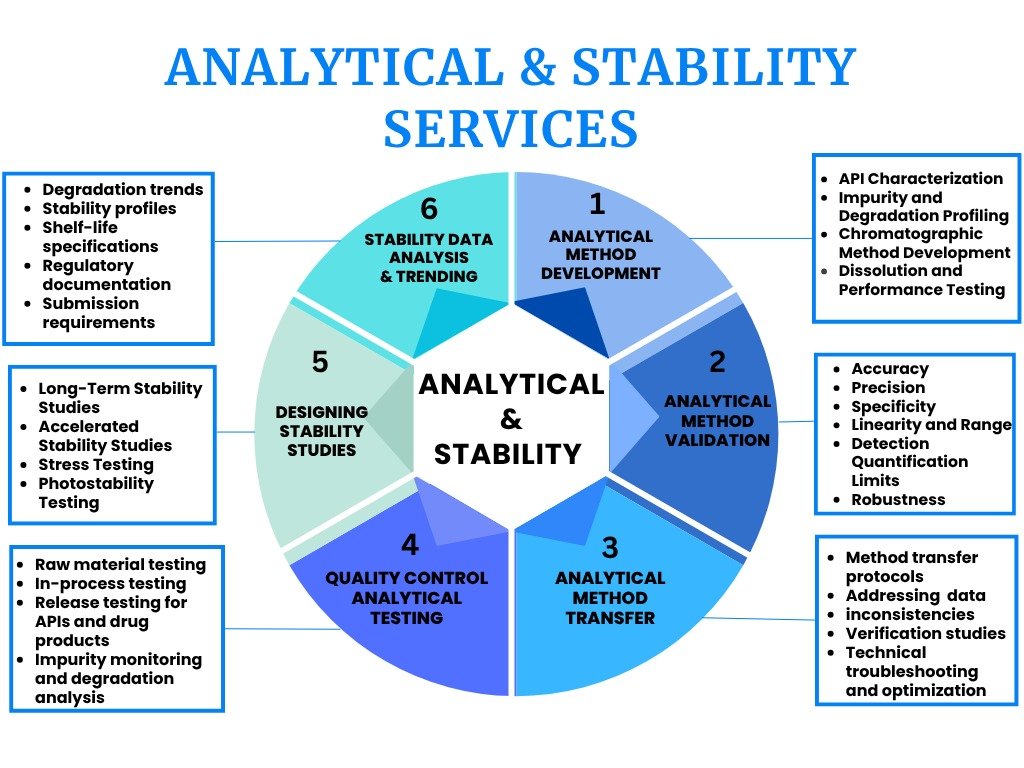
1.Analytical Method Development
The foundation of effective pharmaceutical development starts with competent analytical techniques. At HPC, we create and implement analytical techniques that precisely assess pharmaceutical substances’ identity, purity, potency, and stability. Because each drug molecule possesses distinct chemical traits, necessitating analytical methods that are specially designed for its characteristics. During the entire product period, we create analytical methods that are especially suited for characterizing APIs, intermediates, and pharmaceutical goods.
Prior to implementing analytical approaches, our researchers thoroughly examine the molecular composition, possible contaminants, breakdown routes and formulation properties. This guarantees that the techniques we create are dependable, responsive and indicative of stability.
Our services for method development encompass:
- Characterization of APIs and Development of Assays: We create analytical techniques that precisely measure the strength and concentration of active pharmacological substances. These techniques guarantee accurate drug concentration assessment in both raw materials and final products.
- Profiling of Impurities and Degradation: The impurities, degradation substances and by-products that could arise during production or preservation phases are getting tracked, measured and detected by experts.
- Chromatographic Method Development: We isolate complicated mixtures and guarantee precise quantification of pharmacological agents and other molecules with refined chromatographic procedures like HPLC and GC.
- Testing of Dissolution and Performance: We create dissolution and release testing procedures for pharmaceutical products to assess how the drug performs in physiological environments.
In order to ensure that the resulting analytical processes can accommodate both early development research and commercial production, each technique is built with scalability and regulatory submission in mind.
2.Analytical Method Validation
The process to formulate an analytical technique is merely the initial phase. Analytical techniques must be verified after they are created in order to their dependability, repeatability and appropriateness for regulatory submissions. At HPC, we conduct thorough analytical technique confirmation in accordance with ICH Q2 and various international regulatory standards. Every analytical process operates within predetermined performance parameters in accordance with our validation monitoring.
Our validation research often assesses:
- Accuracy-to verify that the analytical technique measures the analyte’s actual value.
- Precision–to verify that consistency of outcomes under different identical testing circumstances.
- Specificity–to show that the technique evaluates the target substance only free from interference caused by excipients or contaminants.
- Linearity and Range–to confirm that the approach yields proportionate results over applicable concentration ranges.
- Limits of Detection and Quantification–to ascertain the minimum amounts that can be detected and quantifiable concentrations.
- Strong performance–to evaluate how minor conditional changes impact the effectiveness of an analytical approach.
Through the standardized protocols, to validate analytical methods, we guarantee that the produced data is entirely suitable and scientifically trustworthy for regulatory examination by validated analytical procedures.
3.Method Transfer and Lab Validation
The analytical techniques need to be effectively transitioned between labs as pharmaceutical initiatives move from research labs to production facilities while preserving confidential data and reproducibility without neglecting performance. At HPC, we oversee verified method transfer processes in both internal and external labs, notably quality control centers and CDMOs.
Our systematic Method-transfer systematic framework encompasses:
- Developing comprehensive method transfer procedures specifying acceptance necessities and testing circumstances.
- Conducting analysis and comparative testing between laboratories that provide and receive data.
- Examining and addressing inconsistencies in analytical data.
- Verifying that the receiving laboratory can reliably replicate results by performing verification studies.
- Offering technical support and enhancements to address and fix any discrepancies.
Global manufacturing processes and regulatory compliance depend on analytical methodologies operating consistent results across various locations, which is ensured by methodical methodology.
4.Quality Control (QC) Analytical Testing
Ongoing analytical supervision is necessary to guarantee consistency in product quality once it reaches the production site. Ensuring the quality of pharmaceutical products necessitates strict quality assurance tests during both manufacturing and development processes which confirm that APIs, intermediates and final drug products fulfill predetermined equality standards.
At HPC, we offer extensive QC analytical assistance for commercial production activities as well as development programs. Among our quality control testing services encompass:
- Testing of raw materials: Before starting the manufacturing process, we confirm the identity and quality of new raw materials upon arrival to confirm they comply with fully established requirements.
- In-process testing: Throughout production, we oversee key process phases to verify that intermediate and final end products satisfy quality standards.
- Release testing for APIs and pharmaceutical products: Prior to distributing products, we perform comprehensive analytical testing to verify identity, strength, purity, and adherence to regulatory standards prior to distributing products being made available for distribution.
- Monitoring impurities and analyzing degradation: We assess impurity levels to guarantee product dependability and excellence throughout various production batches.
Strict data integrity requirements are adhered to in all QC operations and to check consistency between batches, QC testing is carried out to guarantee recorded analytical methods align regulations.
5.Designing Stability Studies
Analytical testing validates product quality at a particular point in time, whereas stability studies assess how its quality evolves over time. Stability research is carried out to comprehend the performance of pharmaceutical products over time in different conditions like shelf life and storage conditions. At HPC, we use ICH stability recommendations (ICH Q1) to create stability programs aligned with regulations. These research efforts assist pharmaceutical firms in assessing the effects of environmental conditions including temperature, humidity, and light impact product stability.
Our stability study services include:
- Long-term stability Research: We assess pharmaceutical products in suggested storage environments to find out how well pharmaceutical items function over long periods of time.
- Enhanced Stability Investigations: We subject products to higher temperatures and humidity levels to forecast long-term stability over a brief period of time.
- Stress Evaluation: We determine degradation pathways and verify that techniques which can identify possible the degradation products by subjecting compounds to harsh environments.
- Testing for Photostability: To guarantee suitable packaging and storage guidelines, we evaluate the effects of light exposure on medicinal substances.
By implementing meticulously organized stability programs, we produce the scientific information necessary to determine shelf life of product, retest intervals, and storage conditions.
6.Analysis and Trending of Stability Data
Creating stability data is merely one aspect of the procedure. Interpreting and analyzing this data to find patterns, hazards and lasting product behavior is equally crucial. At HPC, we perform thorough analysis and trending of stability data to grasp product performance over time by scientific analysis to detect patterns and possible hazards.
We examine stability information in order to:
- Detect initial deterioration patterns that could affect quality of product.
- Assess stability characteristics from various production batches.
- Define shelf-life requirements based on scientific evidence.
- Assist with submission files and documentation within regulatory demands.
By systematically evaluating methodological data, we convert stability findings into valuable perspectives that direct lifecycle management of product.
Assistance with Regulatory Submissions
In regulatory submissions, Analytical and Stability services are essential in way that regulatory agencies require thoroughly documented and scientifically supported data, product quality, analytical dependability and long-term stability before they are licensing pharmaceutical products.
In order to support regulatory submissions, HPC develop carefully well-organized analytical and stability data packages that include:
- Applications for Investigational New Drugs (IND)
- Applications for New Drugs (NDA)
- Master Files for Drugs (DMF)
- Worldwide regulatory submissions
Our paperwork guarantees that stability assessments, analytical methods and validation information are systematically arranged, supported scientifically and entirely in accordance with international regulatory standards.
The Business Impact: Enabling Scalable and Market-Ready Pharmaceutical Products
We are building a systematic analytical framework for scalable and compliant drug development.
Our offerings assist businesses in achieving:
- Lowered development expenses by means of effective analytical planning.
- Reduce regulatory risk by utilizing verified data packages and completing documentations.
- Strong analytical approaches that assist manufacturing processes.
- Quicker route to market via dependable stability and high-quality information.
HPC assists pharmaceutical researchers in confidently converting promising compounds into products ready for the market by fusing scientific knowledge with regulatory knowledge.
Partner with HPC to Boost your Stability and Analytical Approach
At Hayat Pharmaceuticals Consulting, we assist pharmaceutical firms by providing trustworthy analytical and stability knowledge and expertise to support regulations, product quality and informed development choices. Throughout the product lifespan, our team collaborates closely with clients to develop reliable analytical techniques, produce insightful stability data along with compliance-ready documentation that meets regulatory standards throughout the product lifespan.
We would be happy to work with you if you seek to improve your analytical and stability programs with our skilled scientific assistance.
Get in touch with us:
📞 Phone: +1 (415) 686-8822
✉️ Email: uhayat@hpc-us.com

